Over the last 100 years, our knowledge regarding drug toxicity increased significantly, especially since the thalidomide tragedy. This tragedy, sadly, marked the beginning of the rigorous drug approval and monitoring systems. Therefore, marketed drugs are much safer nowadays. However, there are still many uncertainties regarding the testing of novel drugs, especially considering the existing models, which not always cover all important aspects of the human organism functioning. Unfortunately, omission of some of these aspects still cause tragedies following the actual drug validation process, for example, in the UK in 2006 (TGN1412), and in France not so long ago (2016; BIA 10-2474).
A team of researchers from the University of Maribor drew the attention to potential critical points along the drug validation process to improve the existing process with the aim to avoid similar tragedies in the future. Above all, consideration of the human organism in all specific molecular details has to be in the center of new model development. Because the clinical trials are just the last part of the drug registration process, important steps towards improvement of the models have to be taken much earlier. For that reason, the group suggests improvements on existing in vitro toxicological models, which are in the focus of their paper published in our journal, BJBMS.
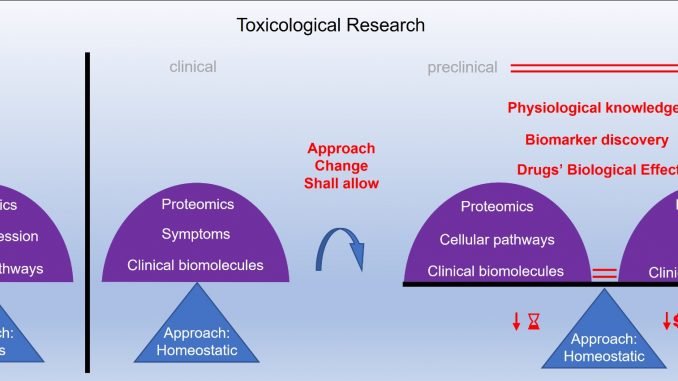
The drug approval process consists of preclinical and clinical testing. However, a huge gap exists between preclinical and clinical research, which prevents continuous monitoring throughout the process. Because of that, the current drug approval process may not accurately predict the drug toxicity. Among the different toxicity models, in vitro cell-based model is found to be the most compatible with clinical models. This model uses all the cell types of the target organ and monitors the cell processes. However, monitoring the maintenance of the cell stability and its internal balance when exposed to different conditions would lead to improvement of the drug validation, because it reflects the biological effects of the drugs more accurately. Furthermore, this leads to better understanding of the human physiology, and would reduce the gap between preclinical and clinical research. In addition, better understanding of drug toxicity further improves assessment and prediction of drug safety.
These improvements may also allow the identification of previously undetectable biomolecules, which may be the potential markers of imbalance of the human organism functioning. “Considering these improvements, we ultimately hope to improve the drug validation process by spanning preclinical and clinical research”, commented Professor Maver.
https://medicalxpress.com/news/2020-01-potential-vitro-toxicological-cell-based.html
http://www.bjbms.org/ojs/index.php/bjbms/article/view/4378
Editor: Edna Skopljak
Leave a Reply